
Description
Measurement of creatinine plays a vital role in diagnosing and treating renal disease. It serves as a valuable tool for evaluating kidney glomerular function and monitoring the effectiveness of renal dialysis. Serum creatinine measurements are particularly valuable in assessing renal health and guiding appropriate medical interventions.
High Values:
Causes:
Kidney failure, congestive heart failure, shock, Heymann’s nephritis, interstitial nephritis, ureterolithiasis, prostatic hypertrophy, desiccation etc.
Low Values:
Causes:
Muscular dystrophy, amyotrophic lateral sclerosis, long recumbency, pregnancy etc.
Features
- High linearity (range 0.1 – 120 mg/dL) and high repeatability
- Little interference by coexisting substances
- Keep the cost down
- Enzyme (SOX-POD) method
Intended Use
Our reagents are specifically designed for the in vitro-quantitative determination of Creatinine in human serum, plasma and urine.
Principle of the Method
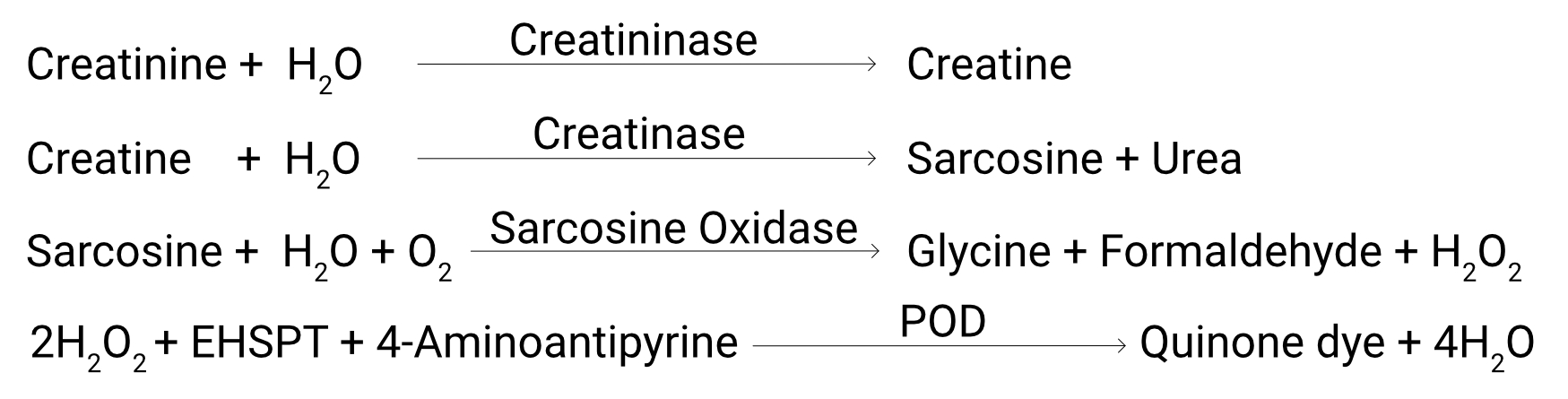
In this enzymatic method, the creatinine present in the sample undergoes hydrolysis, catalyzed by creatininase, resulting in the formation of creatine.
Subsequently, the creatine is converted into sarcosine through the action of creatinase.
The sarcosine is then enzymatically decomposed by sarcosine oxidase, leading to the generation of glycine, formaldehyde, and hydrogen peroxide (H2O2).
The produced H2O2 reacts with EHSPT in the presence of peroxidase (POD), giving rise to a chromophore that is measured at 546 nm. The observed increase in absorbance at 546 nm directly correlates with the creatinine content present in the sample, allowing for quantitative determination.
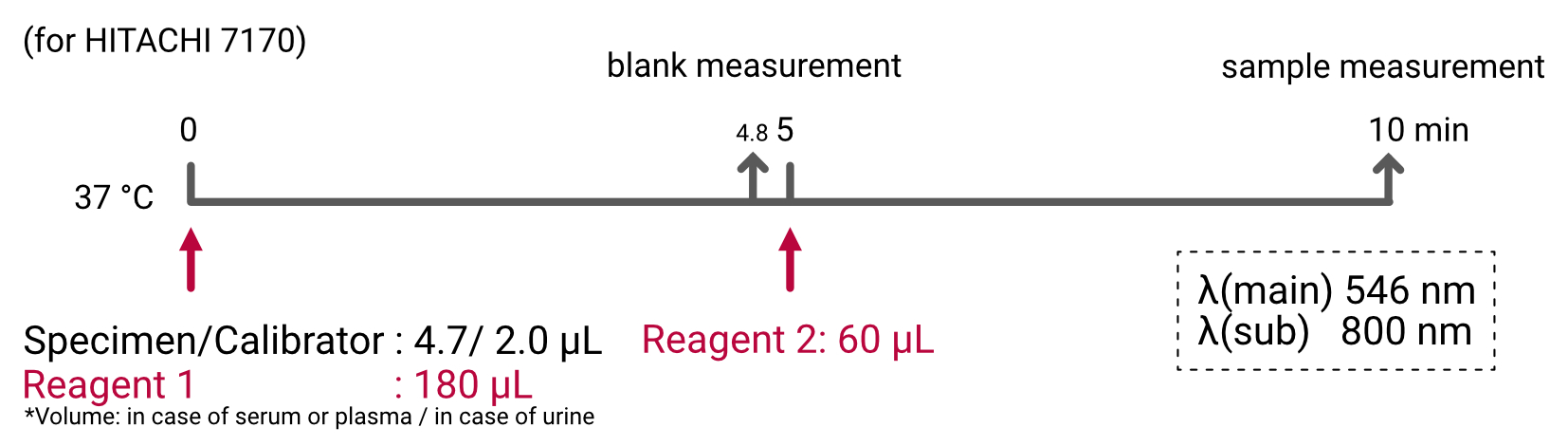
Assay Procedure
Individual instrument applications are available on request.
Assay Reagents, Storage and Stability
Reagent 1: Good’s buffer, Creatinase, Sarcosine oxidase, Ascorbatre oxidase, Catalase, EHSPT
Reagent 2: Good’s buffer, Peroxidase, Creatininase, 4-Aminoantipyrine
Both reagents are liquid, ready to use
Storage: 2-10 °C
Shelf Life: 18 months (before use)
Stability after Opening: at least 2 months at 2-10 °C
Performance Data
The following performance data were obtained on a Hitachi 7170 clinical analyzer.
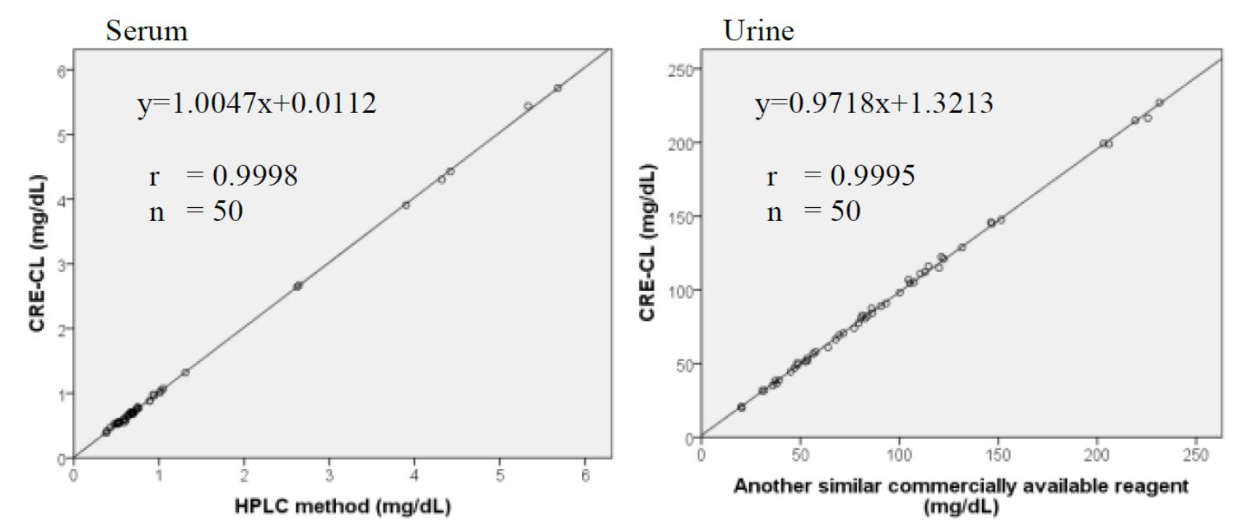
Method Comparison
Comparison studies were carried out using another similar commercially available method. The following results were obtained.
Sensitivity
The sensitivity was evaluated by reading the change in absorbance for purified water sample and serum samples with known concentrations. The results indicated that CRE-CL showed little or no reagent drift on a zero sample.
Under the reaction condition described, 5.0 mg/dL standard solution gives an ABS of 0.063-0.100.
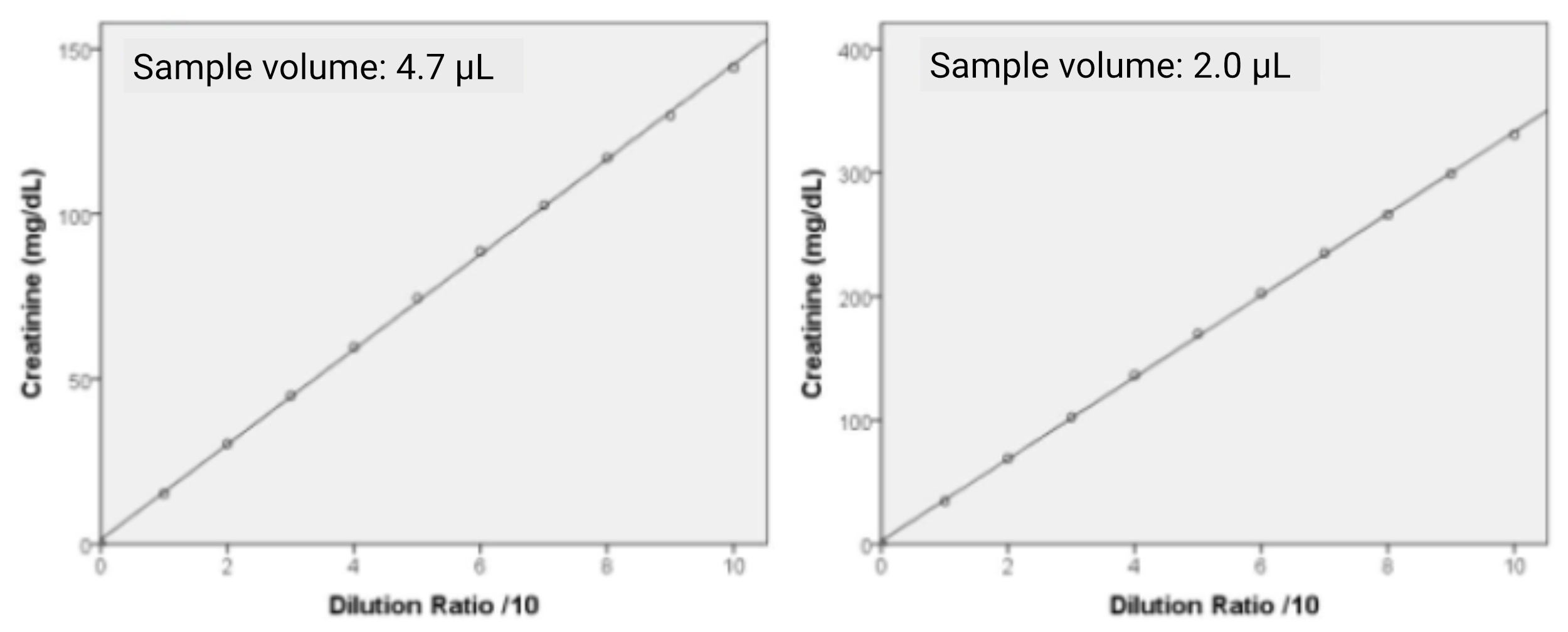
Linearity
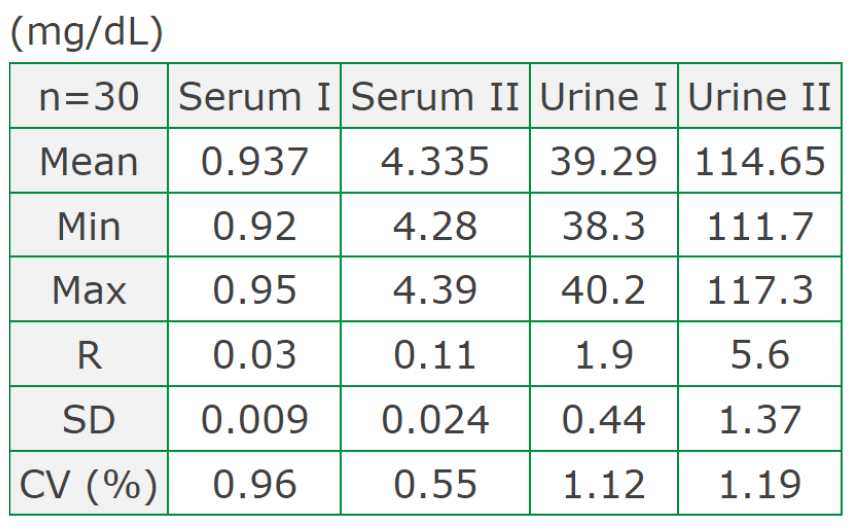
Precision (within-run)
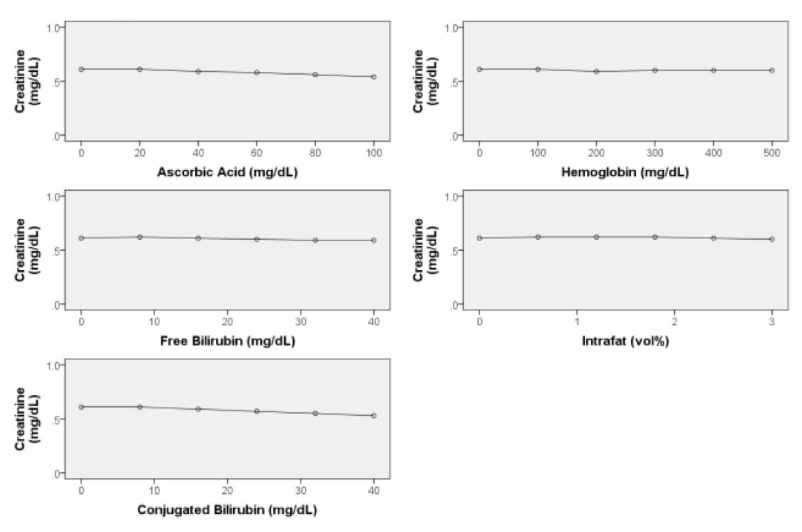
Interfering Substances
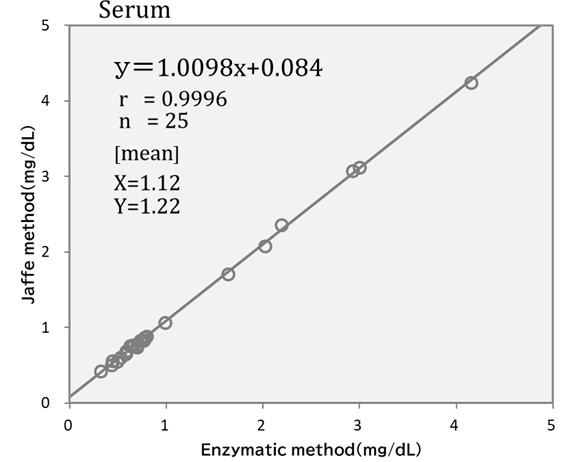
Comparison Data – Enzymatic Method vs Jaffe Method
We performed an comparative accuracy test between the enzymatic method and the Jaffe method by analyzing serum samples treated with creatinine deiminase to deactivate creatinine (serum non-specific reactive test).
The enzymatic method showed an average measurement value of 0.032 mg/dL, while the Jaffe method demonstrated an average measurement value of 0.150 mg/dL. These findings indicate that the enzymatic method displays greater specificity in detecting creatinine.
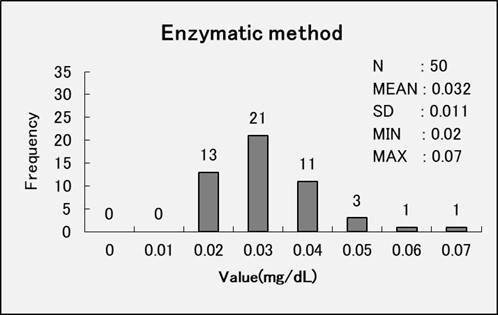
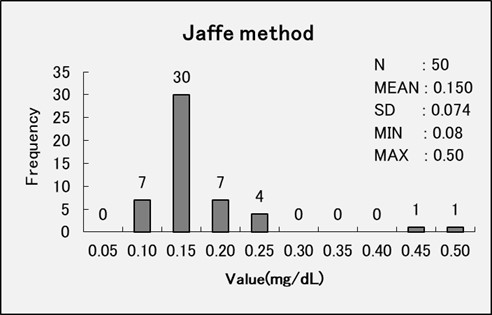
The enzymatic method is a highly specific method for creatinine.
Furthermore, when testing the correlation between the enzymatic method and the Jaffe method, a strong correlation coefficient was observed. However, the results obtained from measuring the same sample showed an average of 1.12 mg/dL, with the enzymatic method, whereas the Jaffe method yielded an average of 1.22 mg/dL, indicating that the Jaffe method produced results approximately 0.1 mg/dL, higher than the enzymatic method. This result aligns with the non-specific reactive test obtained from the “serum non-specific reative test”.
Request Information
Volker Wagner
Sales Manager
