
Description
alpha-Amylase is crucial for the digestion of carbohydrates and is produced by both the pancreas and the salivary glands. Monitoring its levels can offer valuable information about the health of these organs and help diagnose related diseases or conditions, such as acute pancreatitis and other pancreatic disorders as well as mumps and bacterial parotitis.
High Values:
Causes:
1. Pancreatic disease, pancreatitis, trauma, tumours etc.
2. Salivary gland disease, infection, trauma, irradiation
3. Other intra-abdominal condition, biliary tract disease, obstruction, liver disease, mesenteric infarction, acute appendicitis, tumours etc.
Low Values:
Causes:
Exocrine pancreatic insufficiency
Features
- IFCC transferable method
- High linearity (range 3 – 2000 U/L) and high repeatability
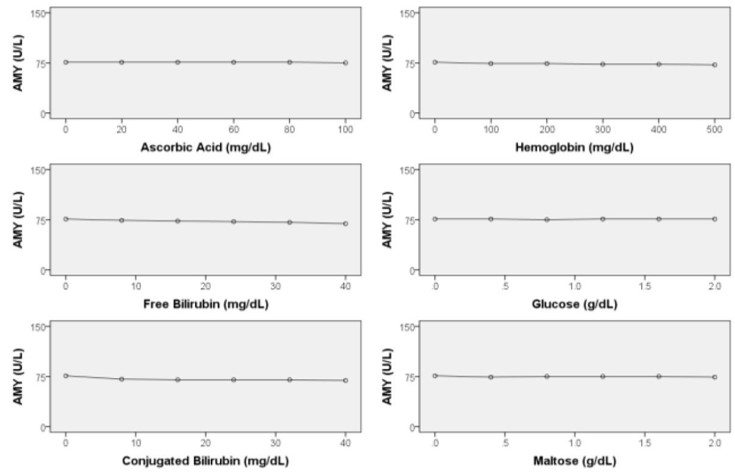
- Little interference by coexisting substances
Intended Use
Our reagents are specifically designed for the in vitro-quantitative determination of amylase (AMY) in human serum, plasma and urine.
Principle of the Method
This method is based on the recommendations of the International Federation of Clinical Chemistry (IFCC).
Ethylidene-4-NP-G7 (Et-4NP-G7) is hydrolyzed by α-amylase to form 4,6-ethylidene-α-(1,4)-D-glucopyranosyl-Gx and 4-nitrophenyl-α-(1,4)-glucopyranosyl-G(7-x). The 4-nitrophenyl-α-(1,4)-glucopyranosyl-G(7-x) is then hydrolyzed into glucose monomers and the 4-nitrophenol by α-glucosidase (α-GH) and glucoamylase (GA). The resulting change in absorbance at 405 nm is proportional to the α-amylase concentration in the sample.
The use of the ethylidene prevents exo – enzymes from breaking down the substrate, so in the absence of α-amylase no color change is observed.
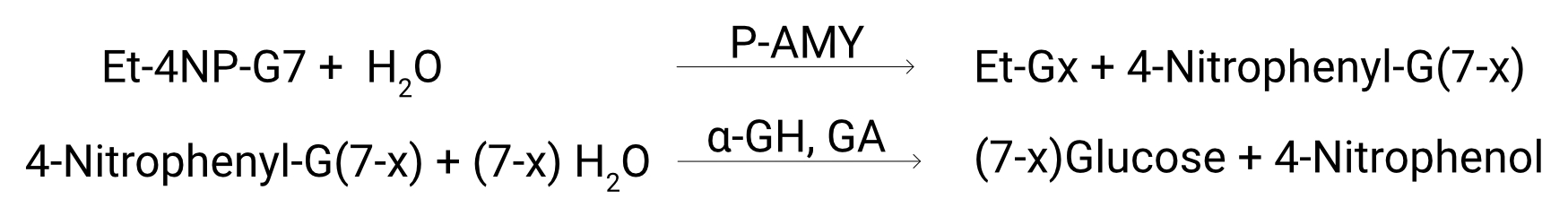
Assay Procedure
Individual instrument applications are available on request.
Assay Reagents, Storage and Stability
Reagent 1: Good’s buffer, α-Glucosidase, Glucoamylase, Sodium chloride, Calcium acetate, pH 7.0 (25 °C)
Reagent 2: Good’s buffer, Et-4NP-G7, Sodium chloride, Calcium acetate, pH 7.0 (25 °C)
Both reagents are liquid, ready to use
Storage: 2-10 °C
Shelf Life: 12 months (before use)
Stability after Opening: at least 1 months at 2-10 °C
Performance Data
The following performance data were obtained on a Hitachi 7180 clinical analyzer.
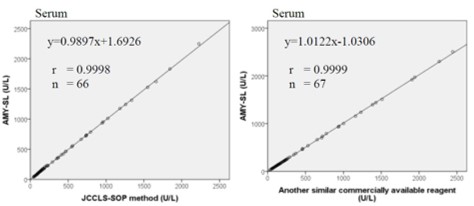
Method Comparison
Comparison studies were carried out using another similar commercially available method. The following results were obtained.
Sensitivity
The sensitivity was evaluated by reading the change in absorbance for purified water sample and serum samples with known concentrations. The results indicated that AMY-SL showed little or no reagent drift on a zero sample.
Under the reaction condition described, 321 U/L AMY activity gives a ΔABS/min of 0.011-0.030.
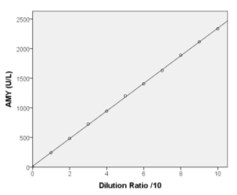
Linearity
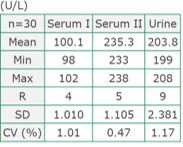
Precision (within-run)
Interfering Substances
Request Information
Aziza Aknin
Key Account Manager
